𝐖𝐡𝐲 𝐕𝐚𝐜𝐜𝐢𝐧𝐞 𝐒𝐚𝐟𝐞𝐭𝐲 𝐌𝐨𝐧𝐢𝐭𝐨𝐫𝐢𝐧𝐠 𝐂𝐨𝐧𝐭𝐢𝐧𝐮𝐞𝐬 𝐋𝐨𝐧𝐠 𝐀𝐟𝐭𝐞𝐫 𝐀𝐩𝐩𝐫𝐨𝐯𝐚𝐥
When a vaccine is approved for public use, many people assume the scientific evaluation is finished.
In reality, approval marks the beginning of one of the most important phases of vaccine safety: post-marketing surveillance.
Modern vaccine systems are designed so that safety monitoring continues long after clinical trials end. Once a vaccine is introduced into real-world use, scientists and regulators gain access to a vastly larger pool of data, millions of people across different ages, health conditions, and environments.
This ongoing monitoring allows researchers to identify extremely rare events, refine recommendations, and ensure vaccines continue to meet the highest safety standards.
It is one of the most important, and often misunderstood, components of modern public health.
Why Monitoring Continues After Approval
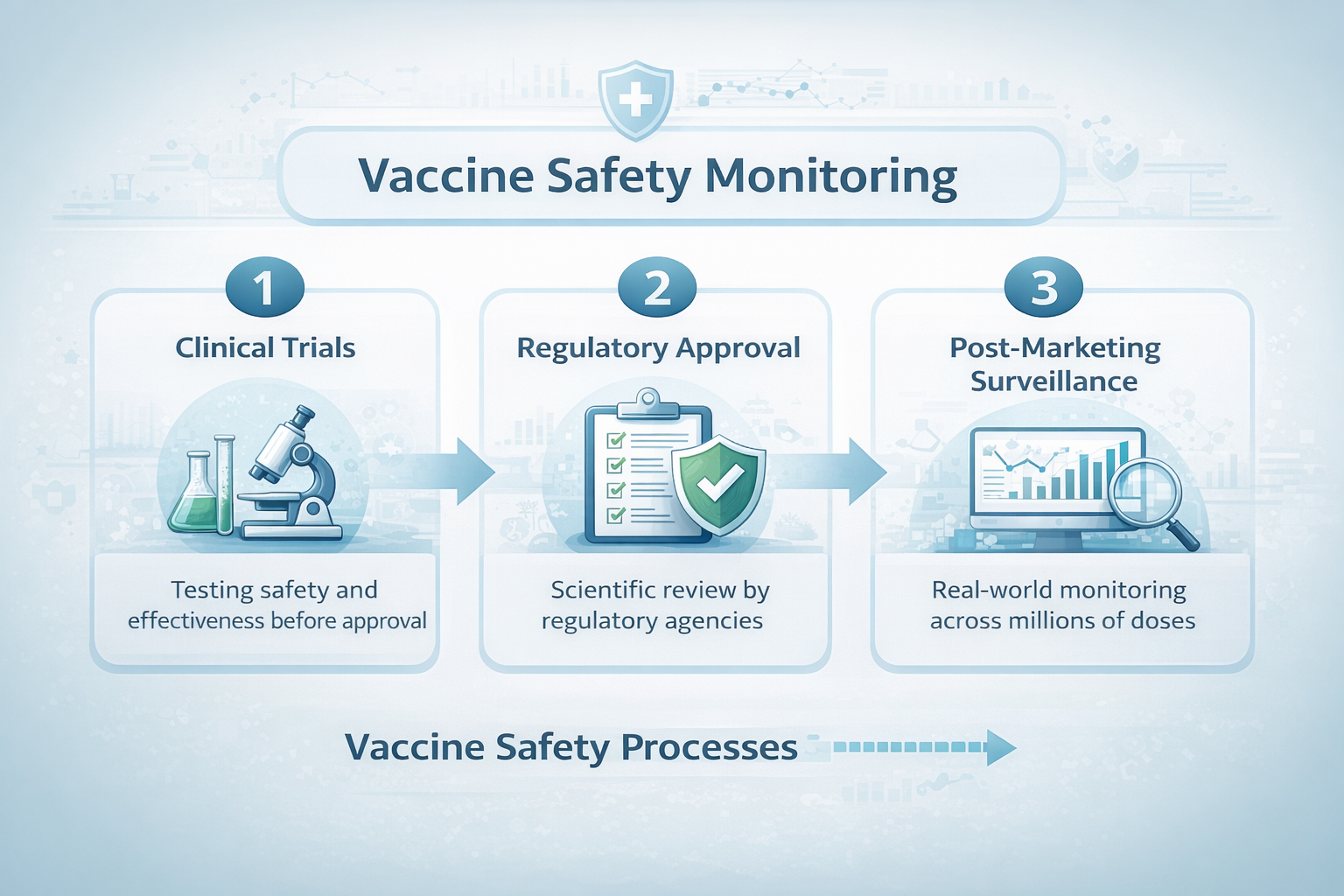
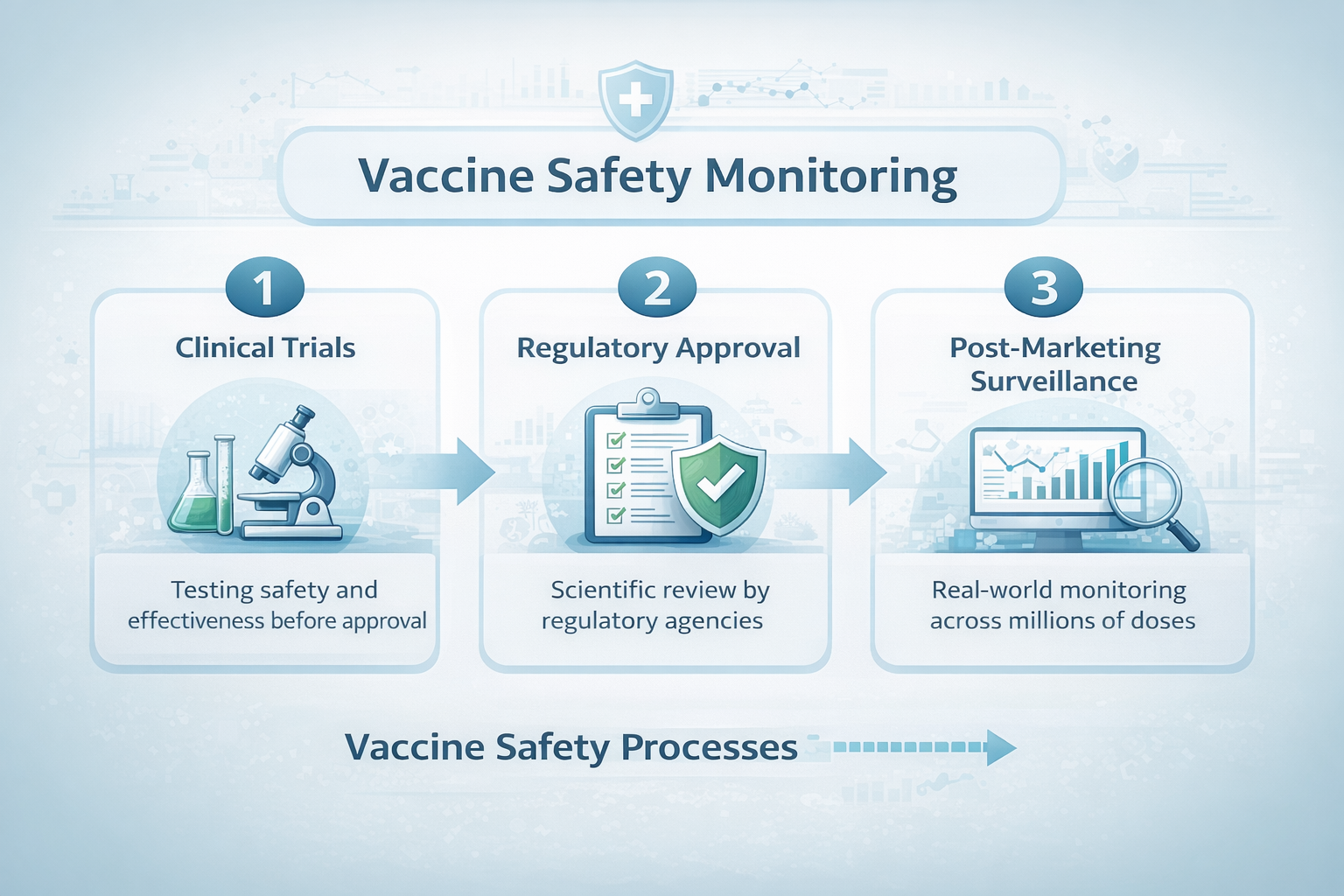
Clinical trials are essential for determining whether a vaccine is safe and effective before it reaches the public. These trials typically involve tens of thousands of participants and are carefully designed to detect common side effects and measure protection against disease.
But even the largest clinical trials cannot capture every possible outcome.
Once a vaccine is introduced into broader use, it may be administered to millions or even hundreds of millions of people. At that scale, researchers can observe how vaccines perform in populations that are far more diverse than any clinical trial can realistically include.
Post-marketing surveillance allows scientists to:
• identify extremely rare adverse events
• monitor vaccine performance across different age groups and medical conditions
• evaluate long-term effectiveness
• refine clinical guidance as new evidence emerges
Rather than signaling uncertainty, this continued monitoring reflects the careful design of modern safety systems.
Approval does not end scientific evaluation, it expands it.
The Systems That Monitor Vaccine Safety
Several complementary systems help track vaccine safety in the United States and internationally. These systems collect and analyze data from clinicians, healthcare systems, researchers, and public health organizations.
Each plays a different role in identifying and evaluating potential safety signals.
Vaccine Adverse Event Reporting System (VAERS)
VAERS is a national reporting system that allows healthcare providers, vaccine manufacturers, and members of the public to report health events that occur after vaccination. These reports do not prove that a vaccine caused the event. Instead, they function as an early signal detection system that helps researchers identify patterns that may warrant further investigation.
Vaccine Safety Datalink (VSD)
The Vaccine Safety Datalink is a collaboration between the CDC and several large healthcare organizations. It uses electronic health records from millions of patients to analyze potential safety signals in near real time, allowing researchers to compare outcomes across vaccinated and unvaccinated populations.
Global monitoring networks
Because vaccines are used worldwide, international regulatory agencies and public health organizations also share safety data. This global cooperation allows scientists to detect potential issues across populations and geographic regions.
Together, these systems create multiple layers of surveillance that make vaccine safety monitoring one of the most comprehensive safety systems in modern medicine.
Why Rare Events Can Take Time to Detect
One reason post-marketing surveillance is so important is that very rare events are difficult to detect during clinical trials.
If a potential side effect occurs once in several hundred thousand or even once in a million doses, it may not appear in pre-approval studies simply because the study population is not large enough.
Large-scale monitoring allows researchers to identify patterns that only become visible when millions of doses are administered.
When such patterns appear, they are carefully investigated to determine whether a causal relationship exists.
This process does not mean the system failed. In fact, it demonstrates the system working exactly as intended.
Scientific understanding evolves as new data becomes available.
How Safety Signals Are Investigated
When monitoring systems detect a potential safety signal, it does not immediately mean a vaccine caused the reported event.
Instead, it triggers a structured scientific review.
Researchers examine multiple forms of evidence to determine whether a true association exists. This may include:
• comparing rates of the event in vaccinated and unvaccinated populations
• reviewing medical histories and underlying health conditions
• evaluating biological mechanisms that could explain the event
• conducting additional epidemiological studies
In many cases, signals are ultimately found to be unrelated to vaccination. In others, the investigation may lead to updated guidance, revised recommendations for certain populations, or additional clinical monitoring.
This careful process ensures that decisions are based on comprehensive evidence rather than isolated reports.
Transparency Strengthens Confidence
The existence of robust monitoring systems reflects a core principle of modern medicine: evaluation does not stop after approval.
Vaccines, like all medical interventions, continue to be studied as new data becomes available. When potential safety signals appear, they are openly investigated and communicated.
Transparency around this process is essential.
When people understand how monitoring systems work — and how potential issues are evaluated — it becomes easier to see that safety is not assumed. It is continually tested.
The Bigger Picture
In public health, trust is built through process.
Post-marketing surveillance is one of the ways that process remains visible and accountable. It ensures that vaccine safety is evaluated not only in controlled trials but also in the complexity of real-world use.
Every dose administered contributes additional data that helps scientists better understand safety and effectiveness.
Approval may mark the beginning of public use, but it is also the beginning of the most comprehensive phase of scientific observation.
In modern medicine, safety is not a single decision.
It is an ongoing process.
And that continuous evaluation is one of the reasons vaccine programs remain among the most closely monitored medical interventions in the world.