Our Blog
Helpful Insights & Updates
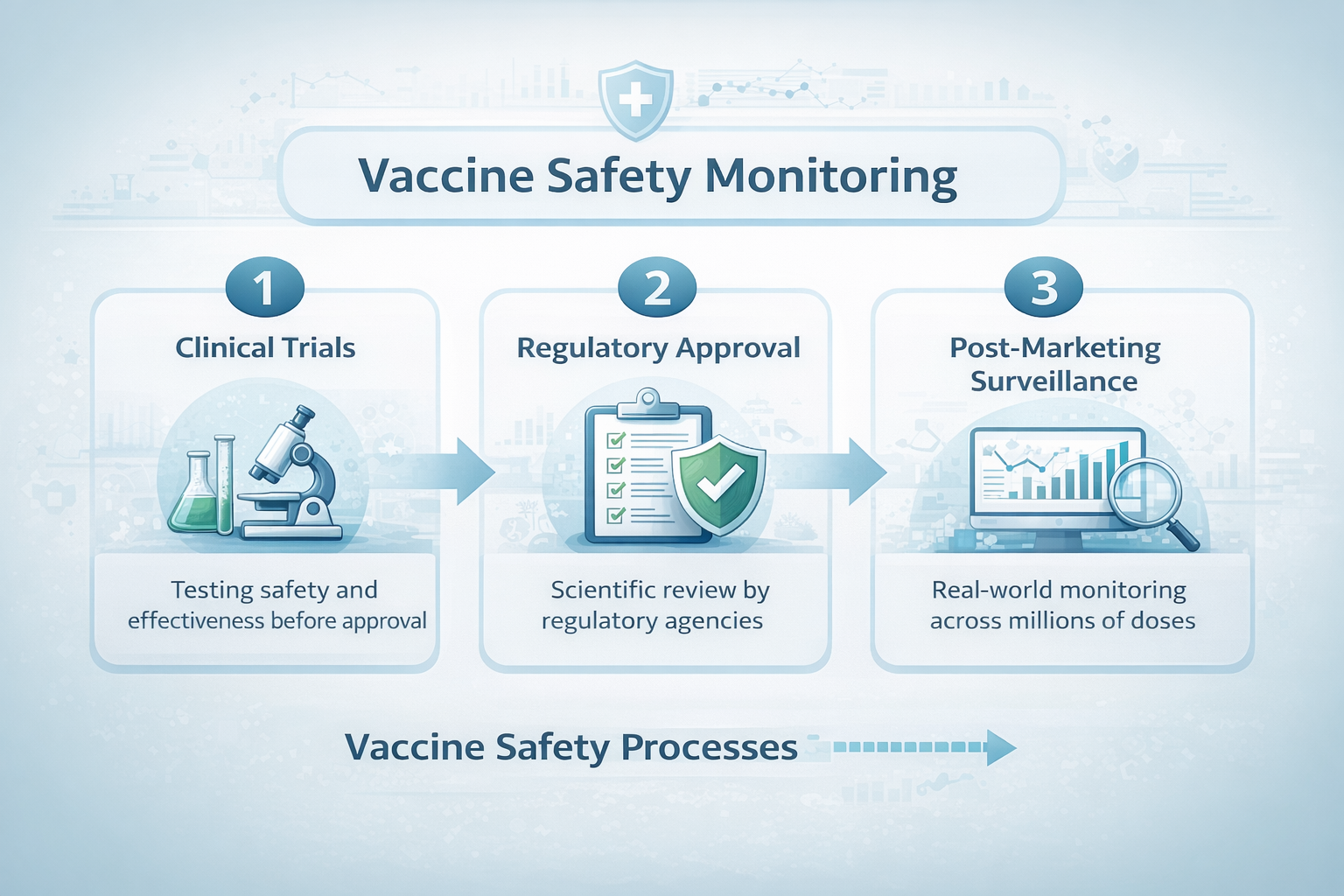
𝐖𝐡𝐲 𝐕𝐚𝐜𝐜𝐢𝐧𝐞 𝐒𝐚𝐟𝐞𝐭𝐲 𝐌𝐨𝐧𝐢𝐭𝐨𝐫𝐢𝐧𝐠 𝐂𝐨𝐧𝐭𝐢𝐧𝐮𝐞𝐬 𝐋𝐨𝐧𝐠 𝐀𝐟𝐭𝐞𝐫 𝐀𝐩𝐩𝐫𝐨𝐯𝐚𝐥
Vaccine safety evaluation continues long after approval through post-marketing surveillance systems that analyze real-world data from millions of people. Learn how monitoring tools like VAERS and the Vaccine Safety Datalink help identify rare side effects, strengthen transparency, and ensure ongoing vaccine safety.
Why Independent Scientific Review Matters in Vaccine Policy
Recently, shifts in federal vaccine advisory processes in the United States have sparked discussion across the healthcare landscape. In response, organizations such as the Massachusetts Medical Society and the Vaccine Integrity Project have announced plans to conduct independent evidence reviews of influenza, COVID-19, and RSV vaccines.
Let’s Bring Better Health to Your Community
Get in touch for fast scheduling, nationwide coverage, and a partner you can trust.